H30+ lewis structure
In the periodic tablehydrogen lies in group 1, and oxygen lies in group Hence, hydrogen has one valence electron and oxygen has six valence electrons.
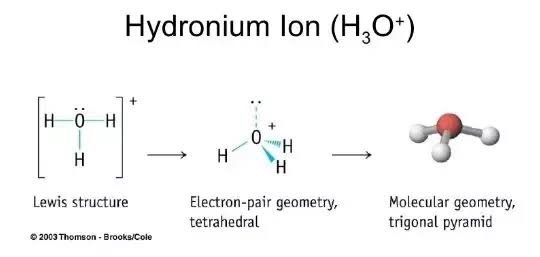
Hydronium ion contains hydrogen and oxygen atoms. Each hydrogen atom has linked with oxygen atom. Only one lone pair exist on oxygen atom. When we draw a lewis structure, there are several guidelines to follow. Number of steps can be changed according the complexity of the molecule or ion.
H30+ lewis structure
There are 3 single bonds between the Oxygen atom O and each Hydrogen atom H. There is 1 lone pair on the Oxygen atom O. Valence electrons are the electrons that are present in the outermost orbit of any atom. Hydrogen is group 1 element on the periodic table. You can see that only 1 valence electron is present in the hydrogen atom as shown in the above image. Oxygen is group 16 element on the periodic table. For selecting the center atom, you have to remember that the atom which is less electronegative remains at the center. Remember: If hydrogen is present in the given molecule, then always put hydrogen outside. You can see the electronegativity values of hydrogen atom H and oxygen atom O in the above periodic table. If we compare the electronegativity values of hydrogen H and oxygen O then the hydrogen atom is less electronegative.
And three O — H bonds are already marked. Here, we have a total of 4 electron pairs.
.
Hydronium ion contains hydrogen and oxygen atoms. Each hydrogen atom has linked with oxygen atom. Only one lone pair exist on oxygen atom. When we draw a lewis structure, there are several guidelines to follow. Number of steps can be changed according the complexity of the molecule or ion. However those all steps are mentioned and explained in detail in this tutorial for your knowledge. There are two elements in hydronium ion; hydrogen and oxygen.
H30+ lewis structure
If we see the nomenclature of hydronium ion, we get to know that according to the IUPAC nomenclature, hydronium ion can be referred to as oxonium. Oxonium is a generalized name for all trivalent oxygen cations, so the use of the name hydronium is necessary to identify hydronium ions particularly. This ion is used in determining the pH of water. The hydronium ion is used in various reactions and the production of different compounds. Both organic and inorganic chemistry includes hydronium ion to a large extent. But before reading the use of this ion in different reactions, we must have knowledge about the basics of this ion, like, lewis structure, geometry, etc.
Uf biology department
Read more about our Editorial process. Remember: If hydrogen is present in the given molecule, then always put hydrogen outside. These outer hydrogen atoms are forming a duplet and hence they are stable. Visit our contact page. Oxygen is group 16 element on the periodic table. Save my name, email, and website in this browser for the next time I comment. Leave a Comment Cancel Reply Your email address will not be published. He has a good conceptual knowledge on different educational topics and he provides the same on this website. Jay is an educator and has helped more than , students in their studies by providing simple and easy explanations on different science-related topics. Only one lone pair exist on oxygen atom.
The Oxygen atom O is at the center and it is surrounded by 3 Hydrogen atoms H. Note: Take a pen and paper with you and try to draw this lewis structure along with me.
He is a founder of Pediabay and is passionate about helping students through his easily digestible explanations. Oxygen is a group VIA element in the periodic table and contains six electrons in its last shell. In order to check the stability of the central oxygen O atom, we have to check whether it is forming an octet or not. Save my name, email, and website in this browser for the next time I comment. This indicates that the oxygen O and hydrogen H are chemically bonded with each other in a H3O molecule. Let me explain the above image in short. If we compare the electronegativity values of hydrogen H and oxygen O then the hydrogen atom is less electronegative. Jay Rana. Because the central atom is bonded with at least two other atoms, and hydrogen has only one electron in its last shell, so it can not make more than one bond. You can see the electronegativity values of hydrogen atom H and oxygen atom O in the above periodic table. Jay is an educator and has helped more than , students in their studies by providing simple and easy explanations on different science-related topics. Scroll to Top.

0 thoughts on “H30+ lewis structure”