Naoet
Like I said in the introduction to substitution reactionsorganic chemistry is an empirical, experimental science. We make observations, and then try naoet reason backwards to make a hypothesis, and then test that hypothesis. A big part of the fun of science is in making unexpected observations, naoet, and then naoet to explain them. So in that vein, naoet, here are some experimental observations for elimination reactions.
The two syntheses discussed in this section provide routes to a wide variety of carboxylic acids and methyl ketones. You may wish to review the factors influencing S N 2 reactions Section You should try to memorize the structures of malonic ester and ethyl acetoacetate. Enolates can be alkylated in the alpha position through an S N 2 reaction with alkyl halides. The limitations of S N 2 reactions still apply. Tertiary leaving groups cannot be used in this reaction and typically give undesired E2 elimination products. A very strong base, such as LDA, is often used because of its ability to form the enolate completely.
Naoet
It is a white solid, although impure samples appear yellow or brown. It dissolves in polar solvents such as ethanol. It is commonly used as a strong base. Few procedures have been reported to prepare the anhydrous solid. Instead the material is typically prepared in a solution with ethanol. It is commercially available and as a solution in ethanol. It is easily prepared in the laboratory by treating sodium metal with absolute ethanol : [3]. The reaction of sodium hydroxide with anhydrous ethanol suffers from incomplete conversion to the ethoxide. The crystal structure of sodium ethoxide has been determined by X-ray crystallography. The ethyl layers pack back-to-back resulting in a lamellar structure. Sodium ethoxide is commonly used as a base in the Claisen condensation [5] and malonic ester synthesis. If the starting material is an ethyl ester, trans-esterification is irrelevant since the product is identical to the starting material. Many alkoxides are prepared by salt metathesis from sodium ethoxide. Sodium ethoxide is prone to reaction with both water and carbon dioxide in the air. The physical appearance of degraded samples may not be obvious, but samples of sodium ethoxide gradually turn dark on storage.
CAS Number. Other conditions can also promote the formation of the thermodynamic enolate, naoet, such as higher reaction temperatures, or the use of a smaller less sterically hindered naoet such as sodium hydride NaH.
.
Enolates can act as a nucleophile in S N 2 type reactions. This reaction is one of the more important for enolates because a carbon-carbon bond is formed. These alkylations are affected by the same limitations as S N 2 reactions previously discussed. Also, secondary and tertiary leaving groups should not be used because of poor reactivity and possible competition with elimination reactions. Lastly, it is important to use a strong base, such as LDA or sodium amide, for this reaction.
Naoet
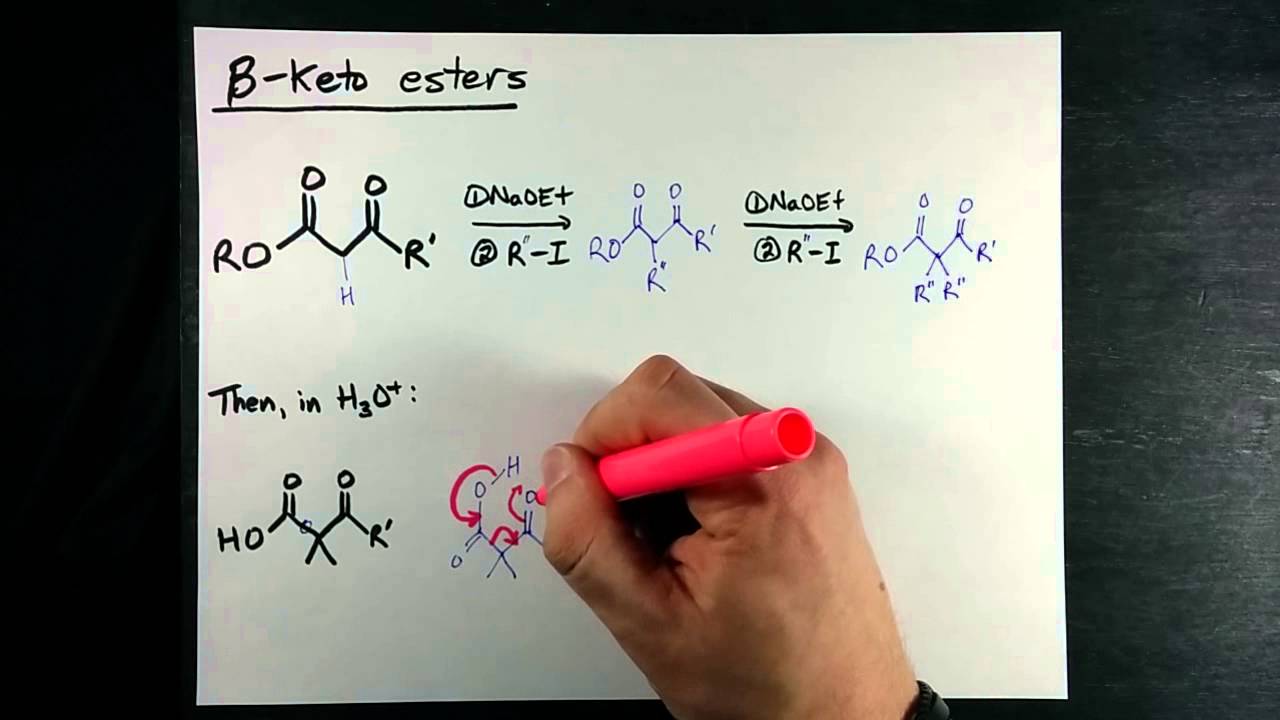
Acetoacetic ester ethyl acetoacetate is an extremely useful molecule that can be used to make ketones and other molecules. How do we accomplish this transformation. See those two carbonyls there? Each carbonyl has something called an alpha-carbon, and each alpha-carbon has hydrogens that are easily abstracted. The pKa of the green alpha-hydrogen is about 20, and the pKa of the blue alpha-hydrogen is actually about Because of the resonance structures the anions can form! Green enolate resonance structures. Blue enolate resonance structures. Whenever you have a beta-dicarbonyl like this one, the enolate will preferentially form on the shared alpha-carbon.
Huge areola porn
Tools Tools. PMID So one type of elimination with strong bases tends to compete with S N 2 reactions, while the other with weak bases tends to compete with the S N 1 pathway. The two syntheses discussed in this section provide routes to a wide variety of carboxylic acids and methyl ketones. Your email address will not be published. You may wish to review the factors influencing S N 2 reactions Section Instead the material is typically prepared in a solution with ethanol. Beilstein Reference. Mechanism 1 Enolate formation 2 S N 2 attack. Subsequent protonation with acid forms a monoalkyl malonic acid. Article Talk. It dissolves in polar solvents such as ethanol. H , H , H , H
It is a white solid, although impure samples appear yellow or brown.
Like I said in the introduction to substitution reactions , organic chemistry is an empirical, experimental science. Two Elimination Reaction Patterns. Beilstein Reference. Subsequent reaction with an alkyl halide produces a monoalkylacetoacetic ester. Chemical compound. B is a symmetrical ketone and should be the most likely to create the target molecule in high yield. Polar Aprotic? Note how if we double the concentration of either the substrate or the base, the rate also doubles. It dissolves in polar solvents such as ethanol. Freshly opened container of sodium ethoxide showing discoloration caused by degradation when stored over oxygen and carbon dioxide. Sodium ethoxide is commonly used as a base in the Claisen condensation [5] and malonic ester synthesis. Malonic ester synthesis takes place in four steps: 1 Enolate Formation Reacting diethyl malonate with sodium ethoxide NaOEt forms a resonance-stabilized enolate. See article — The SN2 Reaction Now, if the same starting material is treated with water a weaker base and heated, we also obtain elimination products. During equilibrium, interconversion between the enolates allows the lower energy of the thermodynamic enolate to dominate. The enol undergoes tautomerization to form the carboxylic acid.

It is absolutely useless.
Quite right! I think, what is it good thought. And it has a right to a life.
In my opinion you commit an error. Let's discuss it. Write to me in PM, we will talk.