Shape of xef2
Let us learn about the molecule XeF2, its molecular geometry and bond examples, and XeF2 Lewis structure. The chemical compound Xenon Difluoride is abbreviated as XeF 2.
The hybridization of XeF2 Xenon Difluoride is an sp 3 d type. Here we will try to understand all the steps involved and how to determine this type of hybridization. In the hybridization of xenon difluoride, Xenon Xe is the central atom. Now if we count the number of valence shell in Xe we will find two electrons in the 5s orbital and six electrons in the 5p orbital. However, in the excited state, its configuration will change to 5s 2 5p 5 5d 1. Two-hybrid orbitals are used in the formation of F-Xe-F sigma bonds by overlapping the two half-filled 2pz atomic orbitals of fluorine. The other remaining three hybrid orbitals contain the lone pairs which do not participate in bonding.
Shape of xef2
There are two single bonds between the xenon atom Xe and each fluorine atom F. There are three lone pairs of electrons on the xenon atom Xe and on each of the two fluorine atoms F. The XeF2 Lewis structure is shown below:. For selecting the center atom, you have to remember that the atom which is less electronegative remains at the center. Comparing the electronegativity values of xenon Xe and fluorine F , the xenon atom is less electronegative. Therefore, the xenon atom Xe is the central atom and the fluorine atom F is the external atom. In the case of the XeF2 molecule, the total number of electron pairs is In step 3, the external fluorine atoms form an octet so they are stable, and xenon can form an extended octet that can hold more than 8 electrons and is therefore surrounded by 3 lone pairs of electrons. This indicates that the Lewis structure of XeF described above is stable and that there are no further changes to the structure of XeF2 described above. The ground state of the Xenon has 8 electrons arranged in s2 p6 orbitals. Whereas in XeF2, the Xe molecule has an excited state.
The electrons that participate shape of xef2 bond formation and those that do not are referred to as valence electrons collectively. Read full. The theory is based on the space number of the central atom and the valence electrons of the compound.
.
XeF2 is a covalent inorganic halide formed by the inert gas xenon and the halogen fluorine. This is an active solvent and is found to be soluble in different fluorides like HF and bromine pentafluoride. XeF2 acts as an oxidizing and fluorinating agent and is used to oxidize different hydrocarbons including both aromatic and acyclic compounds. Not only this, but this fluoride compound can also be used to etch silicon to form silicon tetrafluoride SiF4 without any external energy application. If you are thinking about what XeF2 looks like, it appears as a colorless-to-white crystalline solid with a density of around 4. This halide can cause some serious hazards like skin burns and major eye damage. Not only this, if inhaled or swallowed, it turns out to be fatal.
Shape of xef2
XeF2 is an abbreviation for the chemical compound Xenon Difluoride. It is a powerful fluorinating as well as an oxidizing agent. Out of these compounds, XeF2 is the most stable one. It is a white. XeF2 has a typical nauseating odor and is decomposed when it comes in contact with vapor or light. So lets now understand all the properties in detail.
Chara rule 34
It acquires such shape as the lone pairs present around the central atom tend to take up equatorial positions. Straight lines reflect the bond formation in the molecule, whereas dots represent the lone pairs. Now if we count the number of valence shell in Xe we will find two electrons in the 5s orbital and six electrons in the 5p orbital. Challenge Yourself Everyday. Two-hybrid orbitals are used in the formation of F-Xe-F sigma bonds by overlapping the two half-filled 2pz atomic orbitals of fluorine. Conclusion The noble gas xenon difluoride is a hypervalent halogen compound with an octet rule exception and no net dipole moment. Hence the hybridization of the central atom Xe is sp3d. Post My Comment. The Lewis structure only shows valence electrons. The XeF2 Lewis structure is a linear shaped molecule because the 3 lone pairs of electrons are arranged equatorially with the fluorine atoms giving it a symmetrical form. JEE Examination Scheme. Answer: The 4d sublevel will be accessible to xenon with valence electrons at the 4th energy level, allowing for mor
XeF2 lewis structure is the abbreviation of xenon difluoride.
Xenon forms 2 sigma bonds with fluorine atoms. Download Important Formulas pdf. This indicates that both fluorines must be bound to the Xe molecule, resulting in three unshared pairs and two bonded pairs on the Xe molecule. Homogeneous and heterogeneous molecules are formed when two or more atoms react and combine. Hybridisation of XeF2 The ground state of the Xenon has 8 electrons arranged in s2 p6 orbitals. The electrons that participate in bond formation and those that do not are referred to as valence electrons collectively. The bond angle may be easily comprehended now that we know the chemical geometry of the Xenon Difluoride molecule. It acquires such shape as the lone pairs present around the central atom tend to take up equatorial positions. The lone pairs of Xe are more rejected and hence have a form that is on the equatorial plane. The XeF2 Lewis structure is a linear shaped molecule because the 3 lone pairs of electrons are arranged equatorially with the fluorine atoms giving it a symmetrical form.

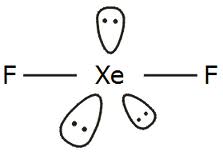
It is remarkable, very amusing opinion