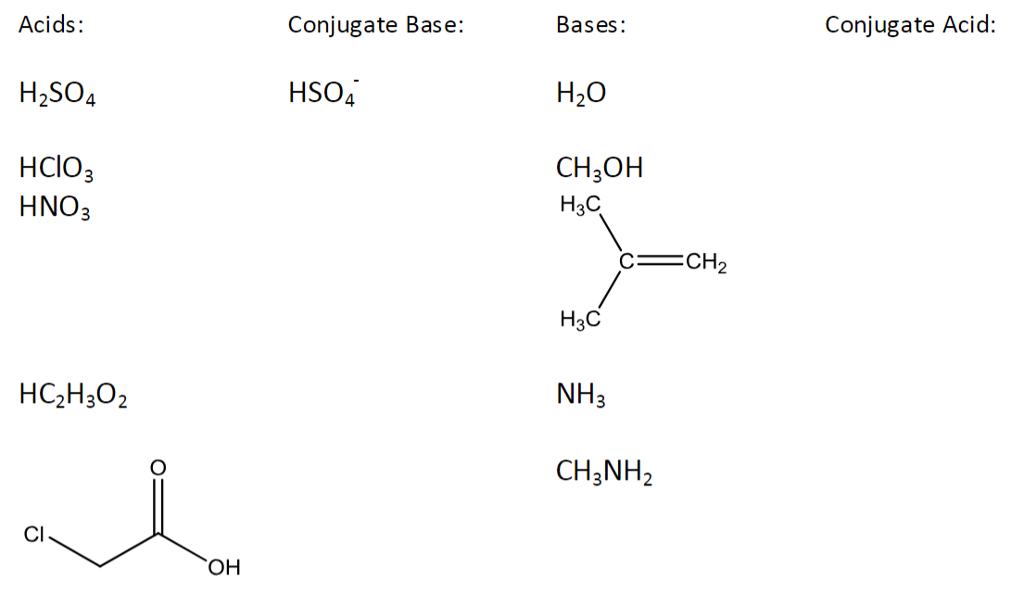
What is the conjugate base of h2so4
Wiki User. They are the products of an acid-base reaction by the Bronsted-Lowry definition.
Identify the acid, base, conjugate acid and conjugate base in the following reaction. HSO4" aq …. A: Acid Base chemistry. Q: Which statement is true of this chemical equation? Q: acid, base, conjugate acid, and conjugate base. Q: Identify the acid, base, conjugate acid, and conjugate base in the following reactions. A: According to Bronsted-Lowry concept an acid is a proton donor and a base is a proton acceptor.
What is the conjugate base of h2so4
What is the conjugate of H 2 SO 4? A non-metallic element is converted into a compound X after a series of reactions. A little amount of X when tested with blue litmus turns to red. X on complete reaction with another compound Y gave the product which did not respond to litmus test. Identify the correct sequence of the reactions. Byju's Answer. Open in App. Conjugate acid and base concept: According to Bronsted Lowry's theory, it is formed by the addition of conjugate acid and conjugate base in a chemical reaction. It is divided into two sections: When an acid is capable of donating a proton and there is an addition of a proton to the acid is known as conjugate acid. When a base is capable of accepting a proton and there is the removal of a proton from the acid is known as conjugate base. For example: The acid-base reaction forms conjugate acid and base by the addition and removal of the proton. Conjugate base: In the above reaction, there is a removal of a proton from the acid and it forms the conjugate base.
Q: The conjugate base of HSO4. X on complete reaction with another compound Y gave the product which did not respond to litmus test.
.
What is the conjugate of H 2 SO 4? A non-metallic element is converted into a compound X after a series of reactions. A little amount of X when tested with blue litmus turns to red. X on complete reaction with another compound Y gave the product which did not respond to litmus test. Identify the correct sequence of the reactions. Byju's Answer.
What is the conjugate base of h2so4
Through examples found in the sections on acids and bases proton-transfer processes are broken into two hypothetical steps: 1 donation of a proton by an acid, and 2 acceptance of a proton by a base. Water served as the base in the acid example and as the acid in the base example [ amphiprotic ]. The hypothetical steps are useful because they make it easy to see what species is left after an acid donated a proton and what species is formed when a base accepted a proton. We shall use hypothetical steps or half-equations in this section, but you should bear in mind that free protons never actually exist in aqueous solution. Suppose we first consider a weak acid , the ammonium ion.
Raileytv onlyfans
Byju's Answer. Q: Consider the following reaction. A: A number of scientists put forward theories to explain the acidic and basic nature of the…. General, Organic, and Biological Chemistry. The material on this site can not be reproduced, distributed, transmitted, cached or otherwise used, except with prior written permission of Answers. What is the base of H2SO4? Q: What is the conjugate base of phosphoric acid? Oxyacids of Phosphorus. For example: The acid-base reaction forms conjugate acid and base by the addition and removal of the proton. Still have questions? See similar textbooks.
On the other hand, a conjugate base is what remains after an acid has donated a proton during a chemical reaction.
The material on this site can not be reproduced, distributed, transmitted, cached or otherwise used, except with prior written permission of Answers. Q: The conjugate base of HSO4. Q: Identify acid, base, conjugate acid, and conjugate base in the following reactio a. Introductory Chemistry: A Foundation. What is the conjugate of H 2 SO 4? The conjugate base of H3O' hydronium ion is a strong acid, strong…. Q: How do you write the conjugate base of each what acids h2so4? H2O… A: conjugate base is formed when an acid donates a proton. Oxyacids of Phosphorus. HCIO4 2.

You are not right. I suggest it to discuss. Write to me in PM.
Completely I share your opinion. In it something is also idea good, agree with you.